
In summary, the key difference between Coulomb's Law and the equation for lattice energy lies in their applications and the interpretation of the distance term. 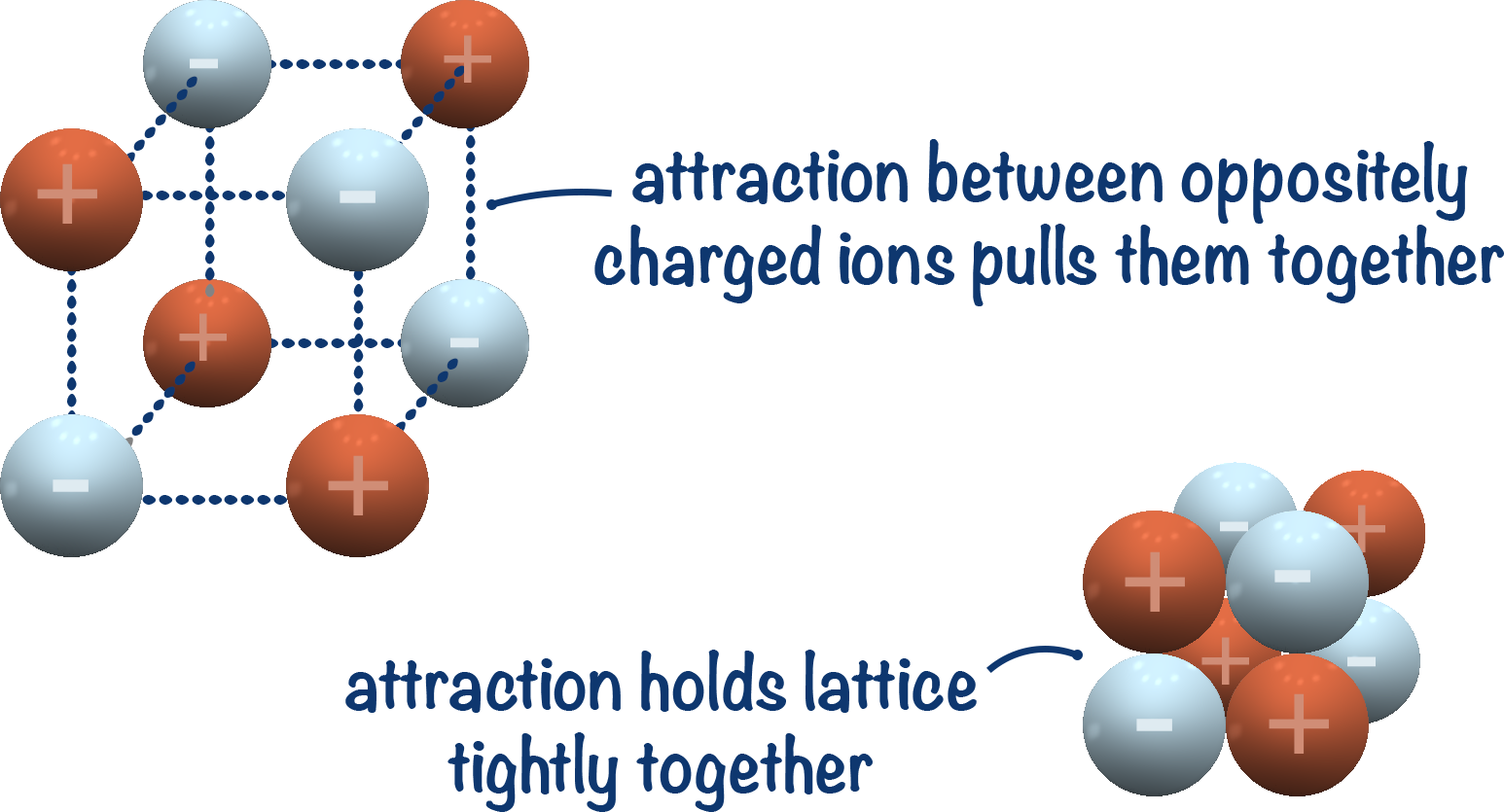
This difference arises because lattice energy represents the energy required to separate ions to an infinite distance, while Coulomb's Law deals with the force between two individual charged particles at a specific distance. Notice that in the expression for lattice energy, the distance "r" is not squared, unlike in Coulomb's Law. The mathematical expression for lattice energy is often given as: Lattice energy depends on the charges of the ions and the distance between them, among other factors. In other words, it quantifies the strength of the ionic bonds that hold the ions together in a crystal lattice. Lattice Energy: Lattice energy is a concept in chemistry that represents the energy required to completely separate one mole of an ionic compound into its constituent ions in the gas phase. Q1 and q2 are the magnitudes of the charges.Ĭoulomb's Law is used to describe how charged particles interact with each other, such as electrons and protons in atoms or ions in ionic compounds. K is Coulomb's constant, a fundamental constant of nature. The mathematical expression for Coulomb's Law is typically written as:į is the electrostatic force between the two charges. It states that the force of attraction or repulsion between two point charges is directly proportional to the product of their charges and inversely proportional to the square of the distance between them. However, they are not the same thing, and the equations involving them can sometimes look similar but have different meanings and applications.Ĭoulomb's Law: Coulomb's Law is a fundamental principle in physics that describes the electrostatic interaction between two charged particles. Lattice energy and Coulomb's Law are related concepts in the field of chemistry, particularly when discussing the forces that hold ionic compounds together. And this is true is look up their values: magnesium fluoride is 2922 kJ/mol while sodium chloride is only 786 kJ/mol. With both of these factors in mind, we should assume that magnesium fluoride has a greater lattice energy compared to sodium chloride. Sodium and magnesium have about the same atomic radii (102 pm and 72 pm respectively), with the main difference being because of the fluoride and chloride ions (133 pm and 181 pm respectively). So magnesium fluoride also has a smaller distance between the ions which also results in greater attraction between the ions and generates a greater lattice energy compared to sodium chloride. The distance between a magnesium and fluoride ion is 205 pm, while the distance between a sodium and chloride ion is 283 pm. 
We can be thorough and also consider the atomic radii of all the ions concerned too. So the product the magnesium fluoride's charges is 2 while the sodium chloride's charge product is only 1. 
This is because the magnesium ion has a +2 charge (and the fluoride has a -1 charge), while the sodium and chloride ions have +1 and -1 charges respectively. If we consider magnesium fluoride (MgF2) and sodium chloride (NaCl), then we would assume that magnesium fluoride would have a greater lattice energy. This means we can usually assume that ions with greater magnitude charges will result in greater lattice energies, and without having to take into consideration the atomic radii. Generally the charges of the ions have more bearing than the distance between them when determining lattice energies. If the goal is to maximize the lattice energy then you'd want ions with larger magnitude charges and/or small in size ions. So both the magnitude of the ion's charges and their atomic radii effect the lattice energy. And the denominator consists of of the distance between the two ions, squared. If we observe the equation of Coulomb's Law the numerator consists of the product of the absolute value of the charges of the ions. So lattice energy is measuring how attracted both the ions are to each other in an ionic compound. In ionic compounds the force is always attractive since the ions have different charges. Whether the force is attractive or repulsive depends on whether the charges have the same sign or not. Coulomb's Law describes the force of attraction (or repulsion) between two point charges. Lattice energies of ionic compounds broadly correspond with Coulomb's Law which Sal provided in the video.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
